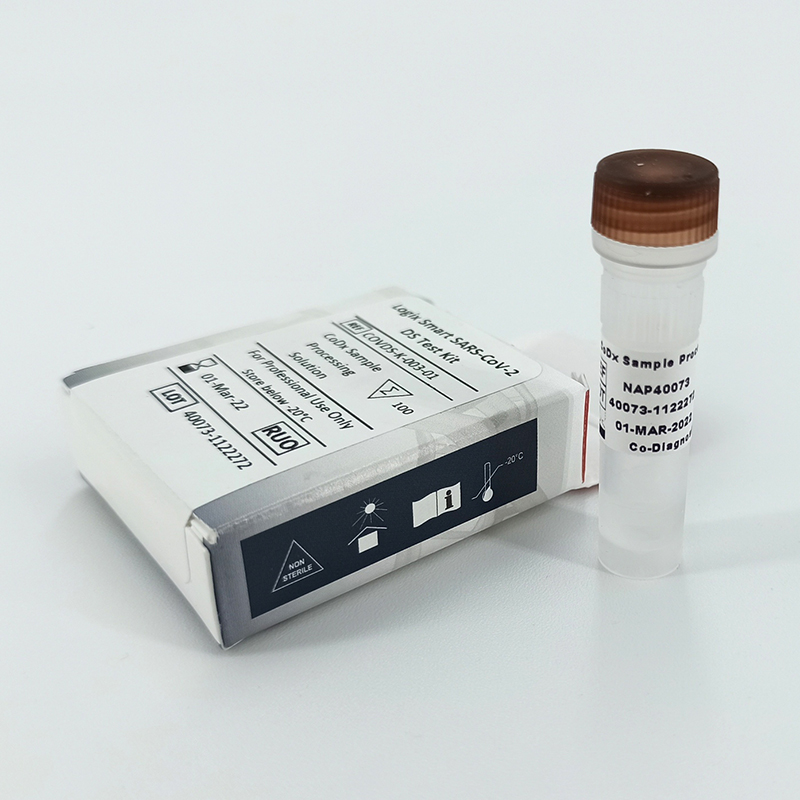
El kit de prueba Logix Smart Coronavirus 2019 (COVID-19) es una herramienta de diagnóstico in vitro innovadora que utiliza la tecnología CoPrimer™ patentada para la detección cualitativa del RNA del coronavirus SARS-CoV-2 (COVID-19). Diseñado para operar mediante un proceso de transcriptasa inversa seguidamente de una reacción en cadena de la polimerasa en tiempo real (RT-qPCR) en una sola etapa, este kit es capaz de analizar fluidos del tracto respiratorio inferior (como lavado broncoalveolar, esputo y aspirado traqueal) y superior (como hisopos nasofaríngeos y orofaríngeos). Es especialmente útil para pacientes que cumplen con los criterios clínicos para la enfermedad del coronavirus 2019 (COVID-19), como establece la Organización Mundial de la Salud (OMS) y los Centros para el Control y la Prevención de Enfermedades de EE. UU. (CDC). El kit incluye un Master Mix listo para usar, completo con un control positivo interno RNaseP para verificar la calidad de la muestra, un control positivo (PC) para verificar el rendimiento de la mezcla maestra y agua libre de nucleasas como control negativo para confirmar que la mezcla maestra está libre de contaminación.
Logix Smart Coronavirus 2019 (COVID-19) | Ref. COVID-K-001 | Presentación: 100 Test/Kit | Marca: Co-Dx
SKU
Pcto-0002
Categorías Aplicaciones, COVID, Diagnóstico enfermedades infecciosas